|
Structure and mutagenesis suggest a different acylation mechanism of ‘substrate-assisted catalysis’ that, unlike other acyltransferases, does not require a proteinaceous catalytic base to complete. Four additional substrate- and product-bound structures uncover the atomic details of its relatively inflexible active site. 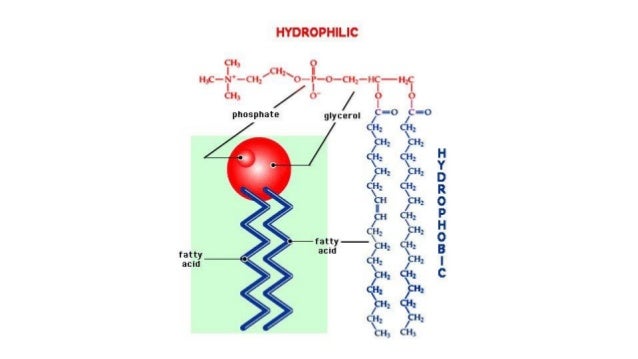
Here we determine the crystal structure of PlsY at 1.48 Å resolution, revealing a seven-transmembrane helix fold. Previous studies have identified several PlsY inhibitors as potential antimicrobials. 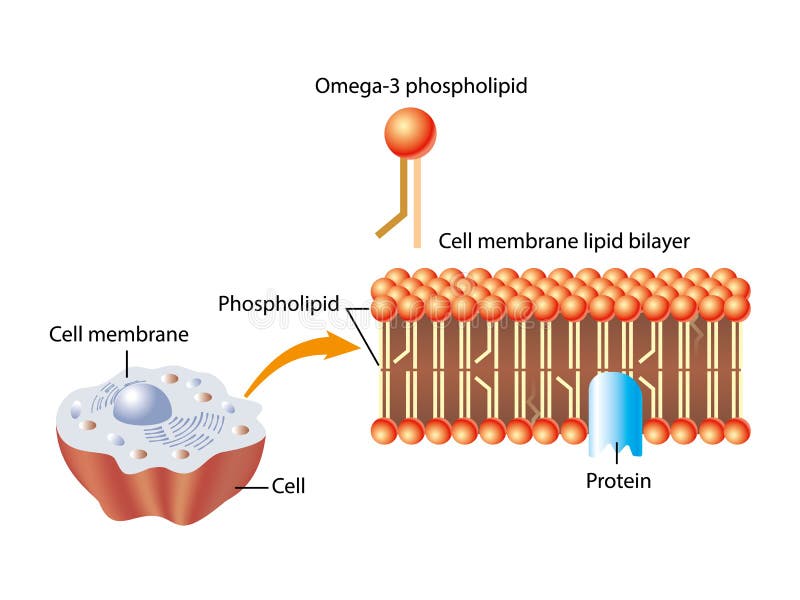
It contains no known acyltransferase motifs, lacks eukaryotic homologs, and uses the unusual acyl-phosphate as acyl donor, as opposed to acyl-CoA or acyl-carrier protein for other acyltransferases. The membrane-integral glycerol 3-phosphate (G3P) acyltransferase PlsY catalyses the committed and essential step in bacterial phospholipid biosynthesis by acylation of G3P, forming lysophosphatidic acid.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
